Outcome measure developed for Crohn's perianal fistula

(HealthDay)—A patient-reported outcome measure (PROM) has been developed for Crohn's perianal fistula, which has good internal consistency, excellent stability, and good responsiveness and construct validity, according to a study published in the September issue ofGut.
Samuel O. Adegbola, M.B.B.S., Ph.D., from Imperial College London, and colleagues generated a draft questionnaire using unstructured qualitative patient interviews on living with Crohn's perianal fistula, a nationwide multidisciplinary consensus exercise, a systematic review of outcomes assessing medical/surgical/combined treatment, and a patient and public involvement day. Psychometric properties, including construct validity, were examined, and reliability and responsiveness were assessed.
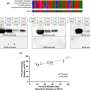
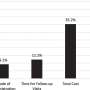
A final 28-item questionnaire was developed using data from 211 patients. The researchers found good internal consistency (Cronbach's alpha, 0.88), excellent stability (intraclass correlation, 0.98), and goodresponsivenessand construct validity for the Crohn's Anal Fistula Quality of Life (CAF-QoL), which showed a positive correlation with the U.K. Inflammatory Bowel Disease Questionnaire and the Hospital Anxiety and Depression Scale.
"Assessing quality of life in patients withinflammatory bowel diseaseis an important component of medical and surgical management and clinical decision making," the authors write. "The CAF-QoL is the first disease-specific PROM in Crohn's perianalfistuladeveloped with a patient-centered methodology."
Several authors disclosed financial ties to the biopharmaceutical industry.
More information:Abstract/Full Text
Copyright © 2021HealthDay. All rights reserved.