寻找炎症的诱因
慢性或急性炎症可导致一系列疾病,其中一些可能是致命的,包括中风、呼吸道和心脏病、癌症、关节炎、哮喘、痴呆、多发性硬化症和糖尿病。今年5月,凯特·劳勒博士和合作者文斯·詹姆斯教授(WEHI)在《科学》杂志上发表了一项研究自然通讯揭示炎症的潜在诱因。
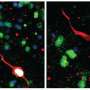
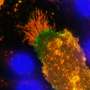
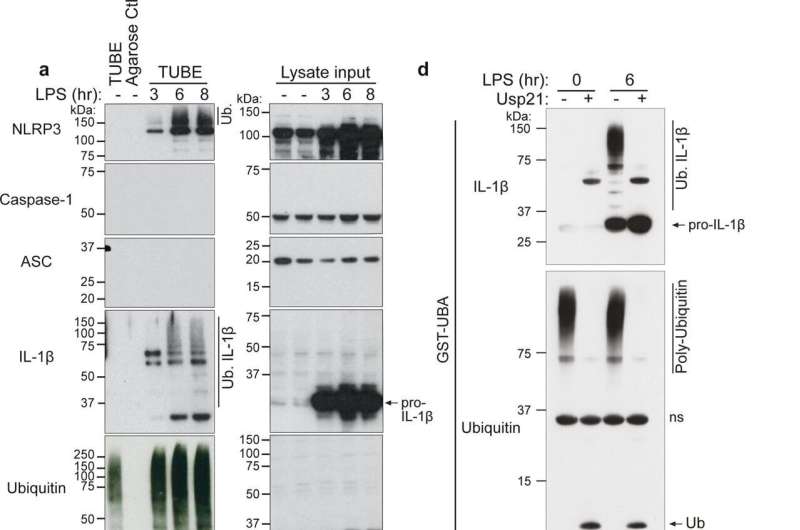
这项研究的重点是细胞因子,白细胞介素-1ß (IL-1ß),它对清除感染至关重要,但也与败血症和驱动自身炎症有关炎性疾病包括风湿性关节炎,2型糖尿病,动脉粥样硬化。
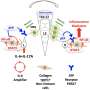
之前的IL-1ß研究主要集中在理解它是如何被触发的,以及抑制这一过程或中和IL-1ß如何减少炎症。然而,很少有人知道前体IL-1ß蛋白是如何被调节的。
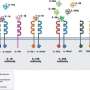
研究小组发现了一个关键事件,该事件有助于非活性IL-1ß的消耗,并限制了激活IL-1ß的酶的获取。发现炎症的潜在触发因素是理解如何操纵IL-1ß水平以限制炎症反应和开发与过度炎症相关疾病的治疗方法的重要一步。
更多信息:Swarna L. Vijayaraj等人,IL-1β的泛素化限制了其被caspase-1切割,并将其作为蛋白酶体降解的目标,自然通讯(2021)。DOI: 10.1038 / s41467 - 021 - 22979 - 3
期刊信息:
自然通讯
引用:寻找炎症的诱因(2022,7月12日)检索自2022年7月18日//www.pyrotek-europe.com/news/2022-07-triggers-inflammation.html
这份文件受版权保护。除为私人学习或研究目的而进行的公平交易外,未经书面许可,不得转载任何部分。内容仅供参考之用。