This article has been reviewed according to Science X'seditorial processandpolicies.Editorshave highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Chances of eliminating HIV infection increased by novel dual gene-editing approach

Gene-editing therapy aimed at two targets—HIV-1, the virus that causes AIDS, and CCR5, the co-receptor that helps the virus get into cells—can effectively eliminate HIV infection, new research from the Lewis Katz School of Medicine at Temple University and the University of Nebraska Medical Center (UNMC) shows. The study, published online in the journalProceedings of the National Academy of Sciences(PNAS), is the first to combine a dual gene-editing strategy with antiretroviral drugs to cure animals of HIV-1.
"The idea to bring together the excision of HIV-1 DNA with inactivation of CCR5 using gene-editing technology builds on observations from reported cures in human HIV patients," said Kamel Khalili, Ph.D., Laura H. Carnell Professor and Chair of the Department of Microbiology, Immunology, and Inflammation, Director of the Center for Neurovirology and Gene Editing, and Director of the Comprehensive NeuroAIDS Center at the Lewis Katz School of Medicine. "In the few instances of HIV cures in humans, the patients underwentbone marrow transplantationfor leukemia, and the donor cells that were used carried inactivating CCR5 mutations."
Dr. Khalili and Howard E. Gendelman, MD, Professor and Chair of the Department of Pharmacology and Experiential Neuroscience at UNMC, were senior investigators on the new study. The two researchers have been long-time collaborators and have strategically combined their research strengths to find a cure for HIV.
"We are true partners, and what we achieved here is really spectacular," Dr. Gendelman said. "Dr. Khalili's team generated the essential gene-editing constructs, and we then applied those constructs in our LASER-ART mouse model at Nebraska, figuring out when to administer gene-editing therapy and carrying out analyses to maximize HIV-1 excision, CCR5 inactivation, and suppression of viral growth."
In previous work, Drs. Khalili and Gendelman and their respective teams showed that HIV can be edited out from the genomes of live, humanized HIV-infected mice, leading to a cure in some animals. For that research, Dr. Khalili and co-investigator, Rafal Kaminski, Ph.D., Assistant Professor at the Center for Neurovirology and Gene Editing at the Katz School of Medicine, combined their expertise in CRISPR gene-editing technology for targeting HIV-1 with a therapeutic strategy known as long-acting slow-effective release (LASER) antiretroviral therapy (ART) that was co-developed by Dr. Gendelman and Benson Edagwa, Ph.D., Assistant Professor of Pharmacology at UNMC. LASER ART holds HIV replication at low levels for long periods of time, decreasing the frequency of ART administration.
尽管LASER-ART能够消除艾滋病毒mice, the researchers found that HIV could eventually re-emerge from tissue reservoirs and cause rebound infection. This effect is similar to rebound infection in human patients who have been taking ART but suddenly stop or experience a disruption in treatment. HIV integrates its DNA into the genome of host cells, it can lie dormant in tissue reservoirs for long periods of time, out of reach of antiretroviral drugs. As a consequence, when ART is stopped, HIV replication renews, giving rise to AIDS.
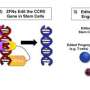
To prevent rebound infection, Dr. Khalili and colleagues began work on next-generation CRISPR technology for HIV excision, developing a new, dual system aimed at permanently eliminating HIV from the animal model. "From success stories of human HIV patients who have undergone bone marrow transplantation for leukemia and been cured of HIV, our hypothesis was that the loss of the virus's receptor, CCR5, is important to permanently eliminating HIV infection," he explained. They developed a simple and more practical procedure for the inactivation of CCR5 that includes an IV inoculation of the CRISPR gene editing molecule.
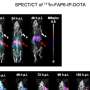
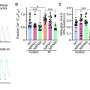
Experiments in humanized LASER-ART mice carried out by Dr. Gendelman's team showed that the constructs developed at Temple, when administered together, resulted in viral suppression, restoration of human T-cells, and elimination of replicating HIV-1 in 58% of infected animals. The findings support the idea that CCR5 has a key role in facilitating HIV infection.
The Temple team also anticipates soon testing the dual gene-editing strategy in non-human primates. To do so, Dr. Khalili will collaborate with Tricia H. Burdo, Ph.D., Professor and Vice Chair in the Department of Microbiology, Immunology, and Inflammation at the Katz School of Medicine, a known expert in the use of non-human primate models for studying HIV-1, who was also a co-author on the new study. Dr. Burdo and her team are interested in understanding the involvement of CCR5 in SIV-infected primates. Her laboratory previously played a key part in research demonstrating the effectiveness and safety of CRISPR-based technology in removing HIV DNA from primate cells.
The new dual CRISPR gene-editing strategy holds exceptional promise for treating HIV in humans. "It is a simple and relatively inexpensive approach," Dr. Khalili noted. "The type of bone marrow transplant that has brought about cures in humans is reserved for patients who also have leukemia. It requires multiple rounds of radiation and is not applicable in resource-limited regions, where HIV infection tends to be most common."
"Curing HIV is the big picture," Dr. Gendelman added. "Through our ongoing collaboration, Temple and UNMC have carried out meaningful research that could ultimately impact the lives of many people."
更多的信息:Dash, Prasanta K. et al, CRISPR editing of CCR5 and HIV-1 facilitates viral elimination in antiretroviral drug-suppressed virus-infected humanized mice,Proceedings of the National Academy of Sciences(2023).DOI: 10.1073/pnas.2217887120